- Products
- Oncohematology
- Antibodies
- Kits
- CAR T-cell
- Euroflow
- Single reagents
- Request info
- Resources and support
- Immunology
- Antibodies
- Single reagents
- Cross match determination (FCXM)
- FcεR1
- Ig subclasses
- Single reagents
- Kits
- TiMas, assessment of tissue macrophages
- Request info
- Resources and support
- Antibodies
- Exosomes
- Accesory reagents
- Software
- Oncohematology
- Services
- Peptide Production
- Design
- Modification
- Protein Services
- Expression and purification
- Freeze drying
- Monoclonal And Polyclonal Antibody Development
- Monoclonal
- Policlonal
- Specialized antibody services
- OEM/Bulk production
- Purification
- Conjugation
- Custom Exosome Services
- Isolation and purification
- Characterization
- Peptide Production
- Shop
- Support
- About Us
- Contact
Exhaustion vs. Dysfunction in CAR-T Cells: Emerging Biomarkers That Are Redefining Their Functional Assessment
CAR-T cell therapy has established itself as one of the most significant advances in modern immunotherapy. In recent years, it has transformed the treatment of numerous hematologic malignancies, particularly refractory leukemias and lymphomas. However, despite its promising clinical results, not all patients achieve durable responses. In many cases, therapeutic efficacy is limited by the progressive loss of function in the modified T cells.
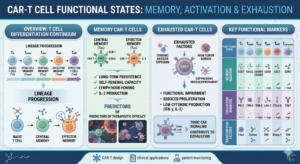
One of the biological phenomena explaining this limitation is the alteration in the functional state of T cells. Traditionally, this problem has been described under the concept of T cell exhaustion, but recent research suggests that the picture is more complex. An increasing number of studies distinguish between T-cell exhaustion and T-cell dysfunction, two related but not identical states that can influence the efficacy of CAR-T therapies in different ways.
Understanding these differences and, above all, identifying reliable biomarkers to detect them has become a priority both for the development of new therapies and for the quality control of cellular products.
The origin of exhaustion in CAR-T cells
T-cell exhaustion is a functional state that arises as a consequence of persistent antigenic stimulation. Under physiological conditions, T cells are activated to eliminate pathogens or tumor cells and subsequently return to a resting or memory state. However, when stimulation persists over long periods of time—as occurs in chronic infections or in the tumor microenvironment—T cells enter an adaptive program characterized by a progressive loss of their functional capacity.
In the context of CAR-T therapy, several factors can promote this process. Continuous exposure to the tumor antigen following infusion is one of the most obvious, but it is not the only one. During the ex vivo manufacturing process itself, T cells may experience intense and prolonged activation signals that contribute to programming states of functional exhaustion even before their administration to the patient. Furthermore, some CAR constructs can generate what is known as tonic signaling, a basal signaling that partially activates T cells even in the absence of antigen.
CAR-T cells that develop exhaustion show a progressive reduction in their cytotoxic activity, lower production of key cytokines, and limited proliferative capacity following infusion. Clinically, this typically translates to reduced persistence of CAR-T cells and an increased risk of tumor relapse.
Exhaustion and dysfunction: two concepts that are not always equivalent
For many years, the terms exhaustion and dysfunction were used almost interchangeably to describe any loss of functionality in T cells. However, deeper molecular characterization of these cell populations has revealed that the two states do not always coincide.
Classic exhaustion corresponds to a relatively well-defined differentiation program, with specific transcriptomic and epigenetic characteristics that develop following chronic antigenic stimulation. This state involves stable changes in gene regulation that determine the cell’s functional behavior.
T-cell dysfunction, on the other hand, can encompass a broader spectrum of alterations, including metabolic defects, problems with intracellular signaling, or states of terminal differentiation that do not necessarily exhibit the classic markers of exhaustion.
This distinction is particularly relevant in the field of CAR-T therapies. Some cells may express inhibitory receptors associated with exhaustion and yet retain significant functional capacity. In other cases, cells that appear phenotypically active may exhibit metabolic or epigenetic defects that limit their therapeutic efficacy.
Classic biomarkers of exhaustion
One of the most widely used methods for identifying states of exhaustion in T cells is the analysis of surface inhibitory receptors, also known as immune checkpoints. Among the most studied are PD-1, LAG-3, TIM-3, TIGIT, and CTLA-4.
Researchers often associate the simultaneous expression of several of these receptors with advanced states of functional exhaustion. In experimental models and clinical studies of CAR-T therapies, they have linked the co-expression of these markers to reduced proliferative capacity and decreased cytotoxic activity.
However, these biomarkers have significant limitations. Many of them are also expressed on recently activated T cells that still retain robust functionality. For this reason, the exclusive use of inhibitory checkpoints can lead to incomplete interpretations of the true functional state of CAR-T cells.
Transcriptomic signatures associated with exhaustion
The development of single-cell transcriptomics technologies has enabled more precise identification of the molecular programs that characterize exhausted T cells. Thanks to these approaches, specific regulatory networks controlled by transcription factors have been described. These networks act as drivers of the exhaustion program. Among the most relevant factors are TOX, NR4A, BATF, and EOMES. Their sustained expression has been associated with the epigenetic programming of this cellular state. In particular, TOX appears to play a central role in reorganizing the chromatin landscape of exhausted T cells.
These transcriptomic analyses have also revealed the existence of intermediate states, such as so-called exhausted T progenitor cells. These populations retain some proliferative capacity and may respond to rescue therapies, such as immune checkpoint blockade.
In the case of CAR-T therapies, the identification of these gene signatures is particularly interesting because some studies suggest that certain predictive transcriptomic profiles are already present before cell product infusion.
Epigenetic biomarkers: stability of the exhaustion program
Beyond changes in gene expression, exhaustion also involves profound modifications in chromatin organization. Studies based on techniques such as ATAC-seq have shown that exhausted T cells exhibit specific patterns of chromatin accessibility. These patterns differ significantly from those observed in effector or memory T cells.
These epigenetic modifications help stabilize the exhausted state. This explains why some therapeutic strategies—such as PD-1 blockade—only partially restore the functionality of these cells. Researchers are exploring epigenetic analysis as a potential tool to assess the functional quality of cellular products. This assessment occurs during the manufacturing process in the field of CAR-T therapies.
The role of cellular metabolism
The functional state of T cells is also closely related to their metabolism. Activated T cells require a dynamic balance between glycolysis, oxidative phosphorylation, and mitochondrial biogenesis to maintain their activity.
Exhausted T cells, in contrast, often exhibit signs of mitochondrial dysfunction, reduced cellular respiration, and alterations in energy generation. These metabolic changes contribute to limiting their proliferative capacity and cytotoxic activity.
For this reason, several biomarkers related to mitochondrial function—such as PGC-1α expression or mitochondrial mass—are being investigated as potential indicators of CAR-T cells’ functional potential.
Memory subpopulations and CAR-T efficacy
Another factor that significantly influences the therapeutic performance of CAR-T cells is the composition of subpopulations within the cell product. Numerous studies have shown that T cells with early memory phenotypes, such as T memory stem cells or central memory T cells, exhibit greater proliferative capacity. They also show higher persistence following infusion. These populations appear to be less prone to developing early exhaustion. This makes them ideal candidates for enhancing the efficacy of CAR-T therapies.
In contrast, cells with terminal differentiation phenotypes typically exhibit limited expansion capacity and a greater predisposition to dysfunctional states.
Toward a multiparametric approach
The growing complexity of the field has led to an emerging consensus: no single biomarker is sufficient to accurately define the functional state of CAR-T cells. Instead, the most promising approaches combine multiple layers of information, including phenotypic, transcriptomic, epigenetic, and metabolic data.
This integrative approach allows for more accurate identification of the cellular states that truly determine therapeutic efficacy. Furthermore, it opens the door to the development of more sophisticated strategies to optimize CAR-T manufacturing and improve their persistence in the patient.
Future perspectives
As CAR-T therapies continue to evolve, the detailed characterization of T-cell functional states is becoming essential in both research and clinical development. Biomarkers of exhaustion and dysfunction will help researchers better understand the mechanisms of therapeutic resistance. They will also enable the design of more robust and durable cellular products.
In the coming years, the integration of multiparametric analysis technologies and artificial intelligence tools is likely to identify predictive signatures of clinical response. This approach will facilitate the personalization of CAR-T therapies. It will also improve their long-term outcomes.