- Productos
- Oncohematología
- Inmunología
- Anticuerpos
- Reactivos individuales
- Determinación de compatibilidad cruzada (FCXM)
- FcεR1
- Subclases Ig
- Reactivos individuales
- Kits
- Solicitar información
- Recursos y soporte
- Anticuerpos
- Exosomas
- Reactivos accesorios
- Software
- Tienda
- Servicios
- Síntesis de Péptidos
- Diseño
- Modificación
- Servicios de Proteínas
- Expresión y purificación
- Liofilización
- Desarrollo de anticuerpos
- Monoclonal
- Policlonal
- Servicios especializados de anticuerpos
- OEM/Producción a granel
- Purificación
- Conjugación
- Servicios de exosomas
- Aislamiento y purificación
- Caracterización
- Síntesis de Péptidos
- Soporte
- Sobre nosotros
- Contacto
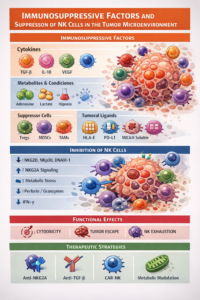
The tumour microenvironment and NK cell suppression: key mechanisms and immunosuppressive
The tumour microenvironment (TME) constitutes a highly dynamic ecosystem in which tumour, stromal and immune cells interact in complex ways. In this context, NK (Natural Killer) cells—essential components of innate immunity with direct cytotoxic capacity—often find their functionality compromised. Understanding the mechanisms of NK cell suppression in the TME is critical for the development of more effective immunotherapeutic strategies.
Role of NK cells in tumour immunosurveillance
NK cells are capable of recognising and eliminating transformed cells without the need for prior sensitisation, thanks to the balance between activating signals (such as NKG2D, NKp30, NKp46) and inhibitory signals (such as KIRs or NKG2A). Under physiological conditions, this balance allows for efficient surveillance against tumour cells with low MHC-I expression.
However, the TME profoundly alters this balance.
Immunosuppressive factors in the TME
Various components of the TME contribute to NK cell dysfunction, both phenotypically and functionally:
1. Immunosuppressive cytokines
- TGF-β: Inhibits the expression of activating receptors (NKG2D, NKp30) and reduces cytotoxicity.
- IL-10: Negatively modulates IFN-γ production.
- VEGF: Interferes with NK cell maturation and recruitment.
2. Metabolites and metabolic conditions
- Adenosine: Generated by CD39/CD73, it inhibits NK activation via A2A receptors.
- Lactate: A product of tumour glycolysis that reduces cytotoxic function.
- Hypoxia: Decreases the expression of activator receptors and alters cellular metabolism.
3. Immunosuppressive cells
- Tregs (regulatory T cells): Secrete TGF-β and consume IL-2, limiting NK activation.
- MDSCs (myeloid-derived suppressor cells): Inhibit cytotoxicity via ROS and arginase.
- Tumour-associated macrophages (TAMs): Polarised towards the M2 phenotype, they contribute to an anti-inflammatory environment.
4. Tumour inhibitory ligands
- Expression of HLA-E → interaction with NKG2A
- Overexpression of PD-L1 → possible indirect impact on NK
- Shedding of NKG2D ligands (MICA/B) → downregulation of receptors on NK
Mechanisms of functional inhibition of NK cells
The above factors converge in multiple mechanisms that impair NK cell function:
- Expression of activating receptors: TGF-β and other factors reduce NKG2D, DNAM-1 and NCRs, diminishing the ability to recognise tumours.
- Inhibitory signalling: Increased ligands such as HLA-E enhance signalling via NKG2A, promoting functional anergy.
- Metabolic alteration: The hypoxic, lactate-rich environment limits oxidative phosphorylation and glycolysis, which are essential for cytotoxic function.
- Decreased cytotoxicity: Reduced release of perforin and granzymes, as well as lower IFN-γ production.
- Functional exhaustion: A phenotype characterised by the expression of immune checkpoints and progressive loss of effector activity.
Therapeutic implications
Detailed understanding of these mechanisms has driven new therapeutic strategies:
- Blockade of NK checkpoints (e.g. anti-NKG2A)
- Neutralisation of TGF-β
- Metabolic modulation of the TME
- Adoptive NK cell therapies (including CAR-NK)
- Combination with monoclonal antibodies and targeted therapies
Conclusion
The suppression of NK cells in the tumour microenvironment represents a significant barrier to effective immune surveillance. The integration of approaches that restore their functionality, alongside therapies targeting the TME, represents a promising avenue for improving outcomes in cancer immunotherapy.